|
Product of rapidly cooled antimony gas, black antimony is amorphous, meaning it has no definite shape. In addition, it may cause headaches, abdominal pain, and may affect the health of the liver, heart, and reproductive system. When inhaled, it causes irritation in the mouth, nose, throat, and lungs. This is a very dangerous process, as antimony trichloride fumes are toxic. /periodic-table-of-the-elements-2017--illustration-769723031-5ac10eb6a9d4f9003769784d.jpg)
When antimony trichloride vaporizes, heat releases, and an explosion of white clouds occur. If antimony scratches the platinum/copper surface, it will result in antimony converting into the more stable metallic allotrope via vaporization. Then, antimony is deposited on the cathode, which can potentially lead to an explosion. In an electrolysis process, there has to be an anode and cathode Antimony is the anode and either platinum or copper is the cathode. First, a concentrated solution of antimony trichloride goes through an electrolysis process in a solution of hydrochloric acid. Electrochemist George Gore was the first to prepare explosive antimony in 1858. It is extremely sensitive, and even a small scratch can trigger a destructive explosion. The explosive allotrope of antimony is a solid solution of antimony trichloride. The metallic allotrope can be thought of as the usual form of antimony. What makes antimony different from other elements is its ability to expand when freezing In total, only four elements can do this, including bismuth. The metallic allotrope is the most stable allotrope of the element antimony. The metastable forms present themselves as yellow, black, and explosive solids. The metallic allotrope is stable, while the three metastable forms are unstable. 
There are four allotropes of antimony: one metallic form and three metastable forms. This element is also one of the heavier pnicogens but is very brittle and bad at conducting electricity. Antimony can either lose the three p electrons, resulting in a 3+ charge, or lose all five electrons to result in a 5+ charge. Additionally, all pnictogens possess five valence electrons, with two of these electrons paired and present in the s subshell, and the remaining three electrons present in the p shell as unpaired electrons. Pnictogens are special because they form strong double and triple covalent bonds (learn more about covalent bonds here) to produce stable compounds.Īntimony can react with almost all of the metals on the periodic table to form pnictides. Along with the element antimony, other elements of this group include nitrogen (learn about the discovery of nitrogen), phosphorus, arsenic, bismuth, and ununpentium. Pnictogen elements are members of the nitrogen group of the periodic table. Antimony has an electron configuration of 4d 105s 25p 3. It lies below arsenic, and above bismuth, and it has properties similar to both of those elements. It lies to the right of tin, and to the left of tellurium. The Element Antimony on the Periodic TableĪntimony, atomic symbol Sb, has atomic number 51 on the periodic table. 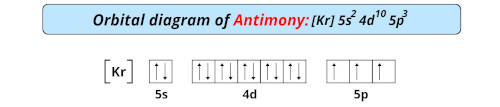
One theory of Wolfgang Amadeus Mozart’s early death is that his doctor poisoned him with a toxic antimony medication.Thankfully, people now know better to not follow these dangerous practices. However, it has been widely used for different medicinal purposes, including laxatives. Antimony is poisonous and should never be inhaled or ingested.There are biblical references of antimony in the Old Testament, where Queen Jezebel uses an antimony compound for makeup.This relates to how antimony was used to make black eye makeup. Stibium originates from the Greek word “stibi”, meaning mark. Antimony’s symbol is Sb, which comes from “stibium”.Its name’s origin comes from the Greek words, “anti” and “monos”, meaning “not alone” because it is always found with another element.Antimony can be thought of as a lonely element because it is never found alone It is always combined with another element. Known since ancient times, this metal has widespread uses, including black eye makeup. This metal has a flaky texture and is hard and brittle. 
The element antimony is a shiny, silver-colored semi-metal of the nitrogen group of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
